Lymphedema Wearable Device Project proves to be a winning collaboration
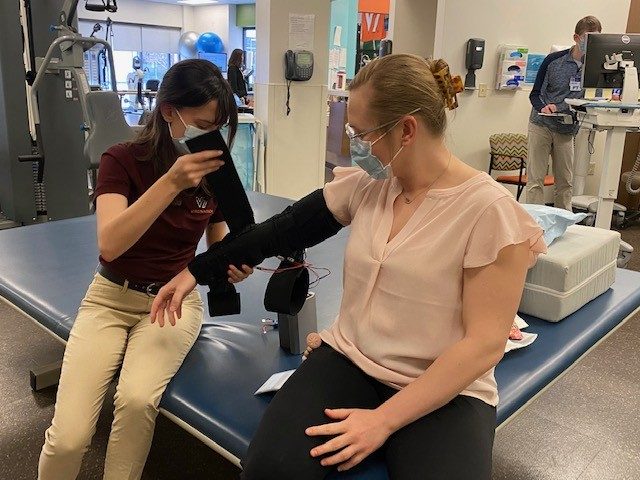
Developed by undergraduate students from the Department of Biomedical Engineering and Mechanics in collaboration with Carilion Clinic’s Innovation team, the device offers patients an in-home treatment option that prioritizes flexibility and quality of life.
Do you recall ever seeing a commercial or advertisement for the treatment of lymphedema? Probably not. It doesn’t have the same name recognition as diabetes or, perhaps, high blood pressure. Yet 3 million to 5 million Americans and close to 200 million people worldwide are affected by the chronic, progressive, and incurable, yet treatable, condition.
Lymphedema, caused by a buildup of lymphatic fluid, is a common side effect of radiation and surgical treatment for cancer patients. It can cause painful swelling, as well as tingling, aching, and fatigue. Current treatments include specific manual lymphatic drainage (MLD) massage and exercises to improve fluid flow. Medical devices, such as lymphedema pumps, are available, but oftentimes they are large, bulky, uncomfortable, stationary, and expensive. Inflexible treatment regimens require clinical visits one to three times a week for hours at a time and can affect patients’ quality of life.
Enter the Lymphedema Wearable Device project.
Based on an idea from Tara Newberry, oncology rehabilitation coordinator at Carilion Clinic, and developed by undergraduate students from the Department of Biomedical Engineering and Mechanics in collaboration with Carilion Clinic’s Innovation team, the device offers patients an in-home treatment option that prioritizes flexibility and quality of life. The motorized compression sleeve uses targeted vibrations mimicking the patterns of MLD and can be controlled by a specialized, downloadable app.

From COVID to competition
During summer 2021, college students around the country were struggling to find internship opportunities because of the global pandemic. Leaning into the idea of collaboration and with funding from the Office of Undergraduate Research, two biomedical engineering and mechanics faculty members approached Carilion Clinic with an idea for a new six-week summer program – the Biodesign Experience for Undergraduates.
Chris Arena, associate professor and director of experiential learning, and Andre Muelenaer, professor of practice, had two primary goals for the new program. First, students would make clinical observations in a hospital setting, seeking out new design opportunities for medical devices. Then, they would work on previously identified ideas for new inventions to improve patient care.
“We always knew we wanted to collaborate with Carilion and had been looking for the right project,” Arena said. “The lack of internships, availability of funding from Virginia Tech, and design concepts created a perfect storm of opportunity.”
Described as “a catalytic agent and resource for innovators,” Carilion Innovation aims to provide resources and support for continuously improving inventions with commercial potential that have been developed through research.
After reading an article seeking projects for the Innovations team, Newberry shared her idea for a mobile, wearable device for at-home use to prolong the effects of massage therapy. Her idea caught the attention and acceptance of the Innovation team, and the lymphedema project began.
With an accepted proposal, a team of undergraduate students, including Seth Jarvis '23, Laura Wenger '23, and Leah Thomas '22, and Ryan McNeil, as well as advisors Arena and Muelenaer, worked to take the project from concept to prototype.
As the program ended, the device showed potential for further development – and Carilion stepped in to sustain the project. Through a Carilion Innovation Proof of Concept grant, which funds and provides project management for high-potential invention, team members received $20,000 toward the development of a prototype and continued work with Newberry. They also added Evgeniya Molotkova, a medical student at the Virginia Tech Carilion School of Medicine, to the team.
Since then, team members have developed engineering abstracts examining the device’s safety profile compared to massages used in the field. They have also successfully demonstrated that the prototype delivers similar results to manual therapy, with the added elements of vibration and different pattern choices to improve fluid flow.

From drawing board to spotlight
In late April, with research, testing, and abstracts behind them, the team was invited to attend the 2023 Design of Medical Devices Conference, hosted by the University of Minnesota, in Minneapolis. It was the first time a student team from the department has earned an invite to the world’s largest medical device conference, giving students an opportunity to present their designs in front of industry professionals.
Over the course of the daylong event, the students answered questions from judges, including executives from Boston Scientific and Medtronic, and shared a device project video.
The team from Virginia Tech took second place in the Five Minute Pitch Competition for medical devices showing the greatest commercial potential. More importantly, the students got a closer look at how novel technologies move from ideation to commercialization.
“We’re at this conference and it’s all these big companies focused on diabetes, cardiovascular, or neurological issues, and it made me realize that without major funding, important projects could die,” said Jarvis. “We aren’t a company. We’re students with a passion and know the work we are doing will ultimately benefit a population that would otherwise not be helped. It feels good to be a part of that kind of change.”

What’s next?
Jarvis and Wenger graduated in May, but the project will continue. New students will join the internship program during the summer and work with Wangler and Molotkova on the lymphedema sleeve. The partnership with Carilion also continues. Next steps include conducting a low-risk feasibility trial, obtaining a full patent and, ultimately, working with a large industry partner to take the product to market.
“The entire project has been a tremendously valuable and successful example of the power of collaboration,” said Arena. “From recognizing a need, partnering with Carilion, and developing a tangible solution while providing students real-life experiences to influence patient care, we couldn’t have asked for a better outcome.”





