Virginia Tech researcher’s discovery may lead to a better understanding of mental health disorders related to stress, anxiety
By studying zebrafish, Albert Pan and his team found a molecule critical in development of the brain’s stress response.
Albert Pan, an associate professor with the Fralin Biomedical Research Institute at VTC, found a connection between a molecule called DSCAML1 and development of the brain’s stress response by studying zebrafish. His findings, published in Frontiers in Cell and Developmental Biology, could lead to a better understanding of stress, anxiety and related conditions in humans. Photo by Clayton Metz.

Virginia Tech researchers studying brain development have identified a molecule that is critical in developing the stress response, a finding that sheds new light on the potential cause of mental health conditions such as stress, anxiety, and related disorders.
Deficiencies in the molecule called DSCAML1, which stands for DS cell adhesion molecule like 1, disrupt brain development and increase baseline levels of cortisol, making the primary stress hormone less able to do its job in times of stress.
The findings — published Thursday, Feb. 16, in Frontiers in Cell and Developmental Biology — point to a better understanding of the role of programmed cell death in the developing brain and may lead to new therapies for stress-related disorders such as depression and anxiety.
“Cell death sounds like a bad thing, but during human development, cell death is important,” said Albert Pan, an associate professor with the Fralin Biomedical Research Institute at VTC.
Programmed cell death rids the body of unneeded cells, such as an excess production of neurons in the brain. Things can go awry if cells die when they shouldn’t or when they fail to die when they should.
Pan’s research team found that the normal process of cell death was disrupted in zebrafish with a DSCAML1 deficiency, indicating the molecule’s role in development of neurons that regulate stress.
The zebrafish is a vertebrate whose genetics have much in common with humans and is an increasingly important model to study brain function and dysfunction that can be applied to humans. In zebrafish, the system that controls stress reactions begins developing within one or two days of fertilization, and stress-induced cortisol signals can be seen within four to five days. The rapid development, combined with the zebrafish’s translucency, gives neuroscientist a window to study the brain.
In the study, some of the zebrafish were genetically modified to interfere with programmed cell death. Scientists focused on the molecule DSCAML1 because genetic changes in its human equivalent are seen in patients with a wide range of mental health disorders, including intellectual disability, autism spectrum disorder, schizophrenia, epilepsy, and stress disorder.
“We found DSCAML1 acts to promote cell death, and cell death is a fundamental mechanism that is involved in many different diseases,” Pan said. Those include not only developmental disorders, but neurodegeneration later in life, as with Parkinson’s and Alzheimer’s diseases. “The more we can understand how the molecule affects normal cell death, the more we can understand how it affects brain function.”
In addition to examining brain development, Pan found changes in the way the zebrafish with the DSCAML1 deficiency responded to stress.
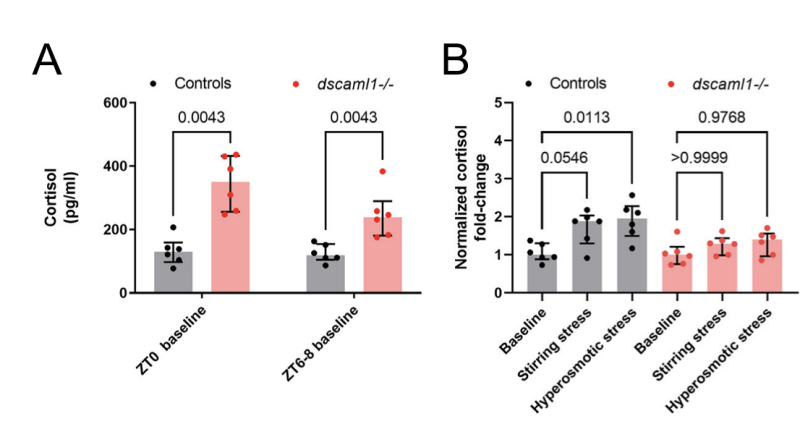
Baseline measurements of the modified zebrafish showed levels of cortisol — the primary stress hormone — two to three times higher than in the control group.
“That is interesting by itself because we still don’t know what is controlling the baseline release of cortisol,” Pan said. “In a lot of psychiatric disorders, there is dysregulation of baseline cortisol release. There are human studies that show there is a link between DSCAML1 regulation and exposure to very stressful experiences.”
A well-functioning stress response can be vital to survival. When faced with a threat, the endocrine, nervous, and immune systems kick into gear. The body becomes more alert, with the brain more clearly focused on actions and behaviors that facilitate survival. There’s also a reduced response to pain from cortisol release and its action on the brain.
Researchers introduced stress by stirring the water in the tanks and changing the water’s salinity, acute stressors the larvae would experience in their natural habitat. In zebrafish with the DSCAML1 deficiency, the response to that stress was out of balance.
“Maladaptive stress responses can cause stress-associated disorders such as anxiety and depression,” said Pan, who also is a faculty member with the Virginia-Maryland College of Veterinary Medicine.
Pan sees the research as opening new lines of inquiry.
“There might be a mechanism where the brain adapts to the environment by adjusting the number of cells, and that might be a significant factor in predisposing people to risk factors later in life,” Pan said.
By determining the significance of cell death, clinicians might be able to better plan therapy using high resolution MRI or other imaging technologies that can resolve groups of specific cell types in the future.
“If there are different ways of treating people who have less – or more – cells, that would be beneficial in terms of targeting the right therapy to the right people,” he said.
Pan received a $2 million, five-year National Institutes of Health grant administered by the National Institute of Mental Health that will allow his lab to further explore the mechanisms that regulate the stress response and the significance of programmed cell death in neurons that coordinate behavioral and endocrine responses to stress. The goal is to develop new therapies for stress-related disorders.
Pan also is the Commonwealth Research Commercialization Fund Eminent Research Scholar in Developmental Neuroscience. His research was supported by the commercialization fund, the Fralin Biomedical Research Institute at VTC, the Virginia-Maryland College of Veterinary Medicine, the Commonwealth Health Research Board, the National Institutes of Health, and Virginia Tech.
The paper’s authors include Manxiu Ma and Kareem C. Clark, formerly of Pan's lab at Fralin Biomedical Research Institute; Alyssa Brunal and Carleigh Studtmann, former Virginia Tech translational biology, medicine and health graduate students; Katelyn Stebbins, a translational biology, medicine and health and Virginia Tech Carilion School of Medicine student; and Shin-ichi Higashijima, with the National Institutes of Natural Sciences in Japan.
When compared with the control group, zebrafish with the DSCAML1 deficiency had elevated baseline cortisol levels and an impaired stress response.